Which of the following is correct for a zero order reaction?
The reverse of this process (the decomposition of ammonia to form nitrogen and hydrogen) is a zero-order reaction. For a zero-order reaction, t12=[A]02k. Thus, as the initial concentration increases, the half-life increases. Hence, graph A is correct.
Which of the following statements is correct i the rate of a reaction decreases with passage of time as the concentration of reactants decreases II the rate of a reaction is same at any time during the reaction III the rate of a reaction is independent of temperature change IV the rate of a reaction decreases with increase in concentration of reactant?
Answer: (a) Rate of reaction is defined as rate of decrease of concentration of any one of reactant with passage of time Thus, as the concentration of reactant decreases with passage of time, rate of reaction decreases.
What are the examples of zero order reaction?
Examples of Zero Order Reaction
- The reaction of hydrogen with chlorine also known as a Photochemical reaction. H2 + Cl2 → 2HCl. Rate = k[H2]0 [Cl2]0
- Decomposition of nitrous oxide on a hot platinum surface. N2O → N2 + 1/2 O2
- Decomposition of NH3 in the presence of molybdenum or tungsten is a zero-order reaction.
What is the unit of zero order reaction?
The units of k for a zero-order reaction are M/s, the units of k for a first-order reaction are 1/s, and the units of k for a second-order reaction are 1/(M·s).
What is a substance that decreases the rate of a chemical reaction called 5 points?
A material used to decrease the rate of a chemical reaction is an inhibitor.
Which of the following is true for the equilibrium constant of a reaction?
Equilibrium reaction means when rate of forward reaction is equal to rate of backward reaction. Hence, we can conclude that out of the given options it is represented by the symbol K, is true for the equilibrium constant of a reaction.
Which statement is wrong about half life period?
And it is a false statement. Half life time is basically defined for the concentration, the time taken to degrade half of the concentration is the half life of the reaction. Hence we can conclude that statements B and D are correct.
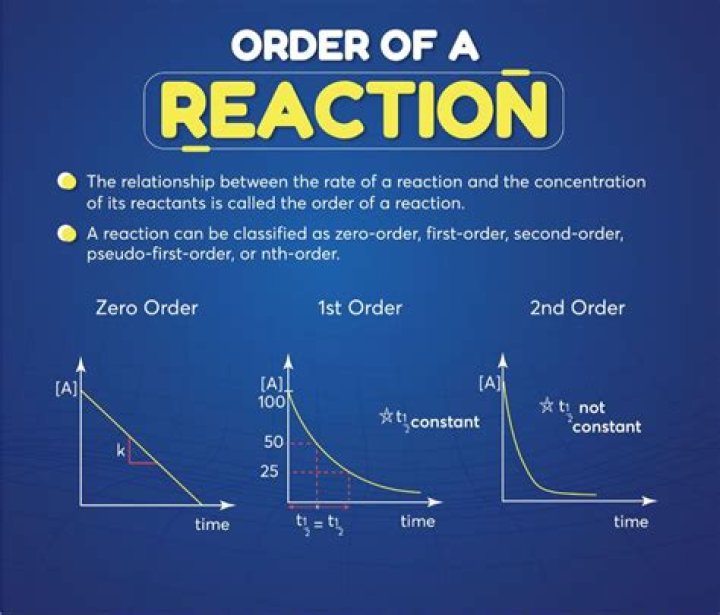
What is the half life period of first order reaction?
The half-life of a reaction is the time required for a reactant to reach one-half its initial concentration or pressure. For a first-order reaction, the half-life is independent of concentration and constant over time.



